.png)
��֢��������չ�ġ�ȼ�ϡ� ����֢����ϵ��3��
.png)
��ͨ����֪����֧�������Ļ�����ʩ�������ġ�������������Ŀǰ��֢�о���һ�����Ż��⡣�ڱ�ϵ�е�ǰһƪ�����У������о��˽�����ϸ�����������������γ�����������Ѫ�ܵġ����죬���ǽ�����һ��ʼ��1863��Ĺ��£���ʱһλ�¹�����ѧ�ң�����³����ά��˹(Rudolf Virchow)����������������

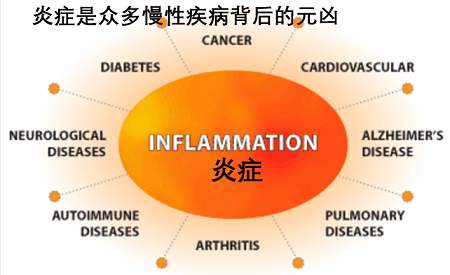
�������������ϵͳ�γ���һ���������ƣ��κ�ս�����Ӷ���Ϊ֮����������ǿ�������֮һ����֢������һ�־��İ��ŵIJ�����ּ������ϸ��������ϸ���ͻ�ѧ�̼���ȵ��ˡ�û���������Ǻܿ��ܻ��Ӥ��ʱ�ڡ�
���ǣ���֢��һ�ַ��ѵ��˸������Զ���Щ���Ҿ�����������ɾ��˺�����������֪������֢�ĺڰ����ǰ�֢��չ��һ��ǿ�����������������ʹٽ���������������ɢ��ȫ����
��������������ŭ
�����Ǵ���֢���������ķ�ʽ��ʼ�����ǵ�Ƥ���ǵ���С�����ߵĵ�һ�����ߡ�����һ��ͻ��������ϰ�������ϵͳ�ķ�ŭ�ͻ��ͷų������������ó�ª������
��ϸ�������������������ʱ������ϵͳ��ϸ��(ͨ������Ϊ����Ѫ��)��嵽���˲�λ���γɴӵ�������ίԱ�ᡣ���ѵ�����ص����������ʼ���������ж��Ļ�ѧ���ʳ�������ߣ������ǵı����ϴ����߰�������������ȥ��
������Ͽ������ַ��ӵ��ƻ����������͡����ȡ��������ʹ�����κ��������˹�ϥ�ǵ��˶�����Ϥ����֢״��
��ܲ��̣����ܿ�ͽ����ˡ����������������Ծ������ٶԽ�����֯�ĸ����������˱��Ե���������ʱ���źŻ��ʹ��ʤ������ϸ�����ش�Ӫ�����ͻָ��Ŷӽ��뵽ָ�����ƵĹ����С�Ѫ�����������γɡ�Ƥ��������������֮�ָ�ƽ����һ���� �ظ�������
��֢���°�֢
���û���������ǿ��������棬���������֢��������ص������ڳ�������֢��һϵ�н�������ı���ԭ�����ʪ�Թؽ���ţƤѢ���ڷ������������е�����ϸ����Rudolf Virchow���������֢�Ƿ�Ҳ�ᵼ�°�֢��
���ҵ��ǣ�������ȷ�ġ�������������֢����(�������Ϳ�����)������һ���˵İ�֢���ա���Ⱦ�Բ�ԭ������İ�֢(�����Ⱦ�����ݸ˾������θ���������Ҹλ���β�����Ⱦ����ĸΰ�)���ص�֮һ��������֢��
�ڶԿ���֢�İ����У�֤��ȷ�䡣
Rudolf Virchow�ǵ�һ������֢�Ͱ�֢��ϵ�������ˡ�
����Խ��Խ���ˡ�
��ô��֢����ε��°�֢����?���ǵ�ǰ��˼����
��һ��С���������������ε�ϸ����ʼ����ʱ�������Դ���Χ�Ļ���������㹻��������Ӫ�����ʡ���������������������ʼ����������һ�п�ʼ��þ�����
���������������棬�����ǻ���Խ��Խ����Ŵ�ȱ��ʱ����ϸ���ͷų�һ�ֻ�ѧ�źţ������źſ�����������Ϊ����ϸ������ϸ��������ϸ������������
һ�����������ڲ�����Щϸ���ͻ����һ�ַ���(��Ϊϸ������)���Ӷ�����Ѫ������(Ѫ������)��������Ҫ��������Ӫ�����ʡ�
����ϸ�����Ӵٽ���һ�ֽ������ʵ�ϸ������ͷ�������������ͬʱ������������ϸ�����÷���(���ɻ�)���ƻ��������Ӷ���һ���ƻ����ǵ�DNA����֢Ҳ����ͨ��������ѧ���ʣ���������ϸ��ͨ������Χ��������õķ��ӣ��Ӷ�����ת�ơ�
�ۺ������������ԣ��տ�ʼ��������ʹ��֢�����������������ٰ�֢�ķ�չ���������ǵ�һλר���������۹���:
��������������ǵ�ȼ����Ļ����ô��֢���ܻ��ṩȼ�ϣ��Ӷ��������档��
������֢
��ô������μ�����֢��? ���������Լ����ڵĿ�ѧ�������о����������֢��ʹ��֢������������������DZ���˿�����֢�ķ��ӻ�·��Ѱ��������һ��ҩ��������ϵͳ�ķ�����
���ǣ�������ǿ���ͨ��������֢��Ԥ����֢�ķ�չ��? ������о�����������𰸺ܿ��ܾ�����һ������ʵ�ϣ���ϣ��������ʼ�����ˡ�
���Ǵ��������֪�ij�Ϊ��˾ƥ�ֵģ�����ˮ�����Ѿ���ʹ����һ����������������֢�������д�����֤����ʾ����Ԥ����֢�����DZ������Ȼ����һ�ַ������Խ��˭Ӧ�÷��ð�˾ƥ�֣����٣��Լ��ʱ�䣬��������ر�������ֹ��֢����δ���İ�֢Ԥ���������з�����Ҫ���á�
³����Virchow��Զ����֪�������о���ȼ��������֢�о�����������������������Ÿոտ�ʼ��
.png)
https://s.click.taobao.com/hYsTINw
��
Feeling the heat �C the link between inflammation and cancer
Category: Science blog February 1, 2013 Safia Danovi3 comments
This entry is part 3 of 5 in the series Microenvironment
Fire_from_loppings
Inflammation is crucial for cancer development
Regular readers will know that the infrastructure supporting a tumour �C its ��microenvironment�� �C is a hot topic in cancer research at the moment. In our previous post in this series, we looked at how otherwise healthy cells collude to form the blood vessels nourishing the tumour. Today, we��ll tell a story that began in 1863, when a German pathologist called Rudolf Virchow peered down his microscope.
Our body��s immune system forms a defensive shield that any fighting force would be proud of. One of its most powerful weapons is inflammation, a carefully orchestrated manoeuvre designed to eliminate enemies such as bacteria, injured cells and chemical irritants. Without it, we probably wouldn��t survive beyond infancy.
But inflammation has a split personality �C one that can wreak havoc for those unfortunate enough to experience it. And we now know that inflammation��s dark side is a powerful force in cancer development, where it aids and abets tumour growth and spread around the body.
Hell hath no fury
Let��s start with the way inflammation normally works. Our skin constitutes the first line of defence against microscopic invaders. But whenever this barrier is breached, the wrath of the immune system is unleashed �C and things get ugly.
As bacteria and other microbes enter the body though an open wound, cells of the immune system (often referred to as ��white blood cells��) rush to the site of injury, forming the welcoming committee from hell. This highly trained militia gets to work immediately, showering intruders with toxic chemicals, punching holes in their surface or swallowing them whole.
From the outside, this molecular thuggery manifests in swelling, heat, redness and pain �C symptoms that anyone who��s ever scraped a knee will be familiar with.
It��s brutal, but it��s over quickly �C it has to be, to minimise collateral damage to healthy tissue. As the enemy is eaten and beaten into surrender, signals urge victorious immune cells to return to base camp. Repair and recovery teams move in to direct the process of healing. Blood vessels sprout. A scab forms. Skin grows. And before long, calm returns and it��s back to business as usual.
The heat is on!
While we might not be able to live without it, too much inflammation can cause serious damage. Chronic, persistent inflammation is behind a host of health problems such as rheumatoid arthritis and psoriasis. And after finding immune cells in tumour samples, Rudolf Virchow was the first to ask whether inflammation might also contribute to cancer.
Unfortunately, he was right �C many chronic inflammatory diseases (such as pancreatitis and Crohn��s disease) can increase a person��s cancer risk. And cancers caused by infectious agents (like stomach cancer caused by infection with the bacteria Helicobacter pylori, or liver cancer caused by infection with the hepatitis B or C virus) are characterised by one thing: chronic inflammation.
In the case against inflammation, the evidence is damning.
Rudolf Virchow was the first to link inflammation and cancer
Rudolf Virchow was the first to link inflammation and cancer
It��s getting hot in here
So how does inflammation lead to cancer? Here��s the current thinking.
When a tiny tumour starts growing from a few rogue cells, it can scavenge enough oxygen and nutrients from its surroundings. But as it grows bigger, demand starts to outstrip supply, and things start getting desperate.
As they struggle to survive, and as they accumulate more and more genetic faults, the cancer cells release chemical signals that lure immune cells called macrophages and granulocytes to infiltrate the tumour.
Once inside the tumour��s inner sanctum, these cells secrete molecules (called cytokines) that kick-start the growth of blood vessels (angiogenesis), which ferry in much-needed oxygen and nutrients.
Other cytokines encourage growth of a sort of cellular ��pillow�� called the stroma against which the tumour rests. Meanwhile, other inflammatory cells spritz the tumour with molecules (free radicals) that further damage their DNA. Inflammation might also fire the starting gun for metastasis by producing chemicals that help tumour cells nibble through the molecules tethering them to their surroundings.
Taken together, it��s clear that fledgling tumours hijack inflammation and use it to accelerate the progression towards full-blown cancer. As one of our own experts once commented:
��If genetic damage is the match that lights the fire, inflammation may provide the fuel that feeds the flames.��
Stay cool, boy
So how do we turn down the heat? Scientists, including our own are working on how to dampen inflammation, making it much harder for cancers to flourish. They��re hacking into the molecular circuitry controlling inflammation, looking for ways to hotwire the system with next-generation drugs.
But what if we could manipulate inflammation to prevent cancer developing in the first place? Recent results suggest that the answer might be anything but next-generation. In fact, it��s been around since Hippocrates.
Better known to most of us by its brand name aspirin, acetylsalicyclic acid has been used for over a century to quell inflammation, and there��s now a body of evidence highlighting its potential in cancer prevention. While there��s still a way to go to work out who should take aspirin, how much, and for how long, it��s becoming clear that blocking inflammation will play a big role in cancer prevention and treatment in the future.
Rudolf Virchow will never know that his work sparked an entire field of cancer research but thanks to him, the fight against cancer is hotting up.
Understanding the link between inflammation and cancer - Cancer Research UK - Science blog http://scienceblog.cancerresearchuk.org/2013/02/01/feeling-the-heat-the-link-between-inflammation-and-cancer/