ĄĄ
p53 and MDM2
ĄĄ
PTEN Protects p53 from Mdm2 and Sensitizes Cancer Cells to chemotherapy.
PTEN (phosphatase and tensin homologue deleted on chromosome 10) phosphatase is the major antagonist of the AKT pathway, it follows that PTEN protects p53 from degradation by inhibiting PI3K and is therefore an important component of the p53 stress response.
DHA/EPA increases PTEN expression which in turn inhibited the NFĶĘB-dependent anti-apoptotic gene transcription
ĄĄ
PTEN Protects p53 from Mdm2 and Sensitizes Cancer Cells to Chemotherapy
https://www.jbc.org/content/277/7/5484.fullĄĄ
ĄĄ
P53 and Mdm2 interaction: How p53 concentration is maintained at low level in normal cells? ~ Biology Exams 4 U
https://www.biologyexams4u.com/2013/08/p53-and-mdm2-interaction-how-p53.html#.XvljNDM6vRgĄĄ
ĄĄ
The MDM2-p53 Interaction
Ute M. Moll and Oleksi Petrenko
Abstract
Activation of the p53 protein protects the organism against the propagation of cells that carry damaged DNA with potentially oncogenic mutations. MDM2, a p53-specific E3 ubiquitin ligase, is the principal cellular antagonist of p53, acting to limit the p53 growth-suppressive function in unstressed cells. In unstressed cells, MDM2 constantly monoubiquitinates p53 and thus is the critical step in mediating its degradation by nuclear and cytoplasmic proteasomes. The interaction between p53 and MDM2 is conformation-based and is tightly regulated on multiple levels. Disruption of the p53-MDM2 complex by multiple routes is the pivotal event for p53 activation, leading to p53 induction and its biological response. Because the p53-MDM2 interaction is structurally and biologically well understood, the design of small lipophilic molecules that disrupt or prevent it has become an important target for cancer therapy.compelling evidence has emerged for MDM2 to have a physiologically critical role in controlling p53.
http://mcr.aacrjournals.org/content/1/14/1001
ĄĄ
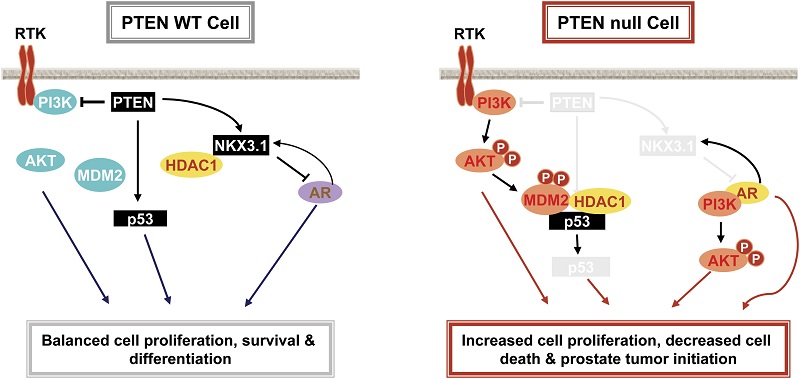
NKX3.1 stabilizes p53, inhibits AKT activation, and blocks prostate cancer initiation caused by PTEN loss
We demonstrate that PTEN loss causes reduced NKX3.1 expression in both murine and human prostate cancers. Restoration of Nkx3.1 expression in vivo in Pten null epithelium leads to decreased cell proliferation, increased cell death, and prevention of tumor initiation. Whereas androgen receptor (AR) positively regulates NKX3.1 expression, NKX3.1 negatively modulates AR transcription and consequently the AR-associated signaling events. Consistent with its tumor suppressor functions, NKX3.1 engages cell cycle and cell death machinery via association with HDAC1, leading to increased p53 acetylation and half-life through MDM2-dependent mechanisms. Importantly, overexpression of Nkx3.1 has little effect on Pten wild-type epithelium, suggesting that PTEN plays a predominant role in PTEN-NKX3.1 interplay. Manipulating NKX3.1 expression may serve as a therapeutic strategy for treating PTEN-deficient prostate cancers.
http://www.cell.com/fulltext/S1535-6108(06)00118-8
ĄĄ
Fish oil targets PTEN to regulate NFĶĘB for downregulation of anti-apoptotic genes in breast tumor growth
Triparna Ghosh-Choudhury,
Abstract
The molecular mechanism for the beneficial effect of fish oil on breast tumor growth is largely undefined. Using the xenograft model in nude mice, we for the first time report that the fish oil diet significantly increased the level of PTEN protein in the breast tumors.In addition, the fish oil diet attenuated the PI 3 kinase and Akt kinase activity in the tumors leading to significant inhibition of NFĶĘB activation. Fish oil diet also prevented the expression of anti-apoptotic proteins Bcl-2 and Bcl-XL in the breast tumors with concomitant increase in caspase 3 activity.
To extend these findings we tested the functional effects of DHA and EPA, the two active ĶØ-3 fatty acids of fish oil, on cultured MDA MB-231 cells. In agreement with our in vivo data, DHA and EPA treatment increased PTEN mRNA and protein expression and inhibited the phosphorylation of p65 subunit of NFĶĘB in MDA MB-231 cells. Furthermore, DHA and EPA reduced expression of Bcl-2 and Bcl-XL. NFĶĘB DNA binding activity and NFĶĘB-dependent transcription of Bcl-2 and Bcl-XL genes were also prevented by DHA and EPA treatment. Finally, we showed that PTEN expression significantly inhibited NFĶĘB-dependent transcription of Bcl-2 and Bcl-XL genes.
Taken together, our data reveals a novel signaling pathway linking the fish oil diet to increased PTEN expression that attenuates the growth promoting signals and augments the apoptotic signals, resulting in breast tumor regression.
Keywords: PTEN, NFĶĘB, DHA, EPA, Breast tumor growth, Apoptotic signal
Fish oil targets PTEN to regulate NFĶĘB for downregulation of anti-apoptotic genes in breast tumor growth
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2847452/
AĄĄ
The Role of MDM2 in Promoting Genome Stability versus Instability
In cancer, the mouse double minute 2 (MDM2) is an oncoprotein that contributes to the promotion of cell growth, survival, invasion, and therapeutic resistance. The impact of MDM2 on cell survival versus cell death is complex and dependent on levels of MDM2 isoforms, p53 status, and cellular context.Extensive investigations have demonstrated that MDM2 proteinĻCprotein interactions with p53 and other p53 family members (p63 and p73) block their ability to function as transcription factors that regulate cell growth and survival. Upon genotoxic insults, a dynamic and intricately regulated DNA damage response circuitry is activated leading to release of p53 from MDM2 and activation of cell cycle arrest.
What ensues following DNA damage, depends on the extent of DNA damage and if the cell has sufficient DNA repair capacity. The well-known auto-regulatory loop between p53-MDM2 provides an additional layer of control as the cell either repairs DNA damage and survives (i.e., MDM2 re-engages with p53), or undergoes cell death (i.e., MDM2 does not re-engage p53). Furthermore, the decision to live or die is also influenced by chromatin-localized MDM2 which directly interacts with the Mre11-Rad50-Nbs1 complex and inhibits DNA damage-sensing giving rise to the potential for increased genome instability and cellular transformation.
Keywords: MDM2; p53; DNA damage; genome instability
IJMS | Free Full-Text | The Role of MDM2 in Promoting Genome Stability versus Instability
https://www.mdpi.com/1422-0067/18/10/2216ĄĄ
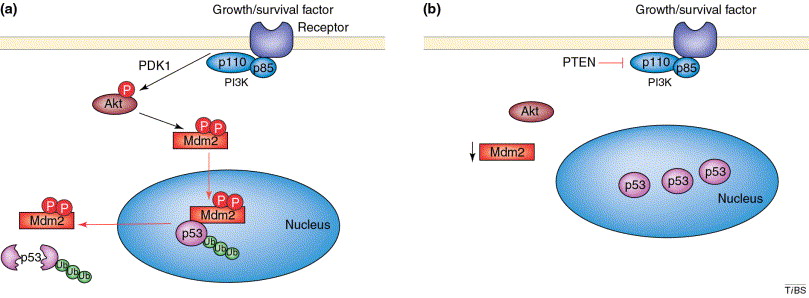
The PTEN, Mdm2, p53 tumor suppressorĻConcoprotein network
Oncoproteins and tumor-suppressor proteins regulate cell growth and viability. Recent observations show that phosphoinositide 3-kinase (PtdIns 3-kinase)ĻCAkt signaling promotes the phosphorylation and movement of the Mdm2 oncoprotein into the nucleus, where it downregulates the p53 tumor-suppressor protein. The PTEN tumor suppressor protein inhibits activation of Akt and this restricts Mdm2 to the cytoplasm. Restriction of Mdm2 to the cytoplasm promotes p53 function and thereby sustains the sensitivity of cancer cells to chemotherapy. p53 acutely induces Mdm2, providing damaged cells the opportunity for repair, but subsequently induces PTEN, favoring the death of mutated or irrevocably damaged cells. Thus, oncoproteins and tumor suppressor proteins are networked to promote normal cell function and eliminate mutated cells.ĄĄ
http://www.cell.com/trends/biochemical-sciences/fulltext/S0968-0004(02)02166-7
ĄĄ
.gif)
.jpg)
.jpg)