��
��֢��ϸ��������л���쳣
Dysregulation of iron metabolism in cancer stem cells ...
Department of Biomedical Sciences for Health, University of Milan, Via
Mangiagalli 31, 20133 Milano, Italy
ǿ��
• ��֢��ϸ����CSCS���������ھ��и�ϸ����������ϸ����
• ������ΪCSCs��������������������ת�ƺͻ�ѧ��ҩ�ԡ�
• ���CSC���Ʒ��ķ�չΪ���ư�֢���Ʊ���ϣ����
• ����̬��ʧ��������֢��ϸ���Ĺ�ͬ������
• ��������Ŀ��CSC��Ҫ���õ��˽�CSC�е�����л��
����
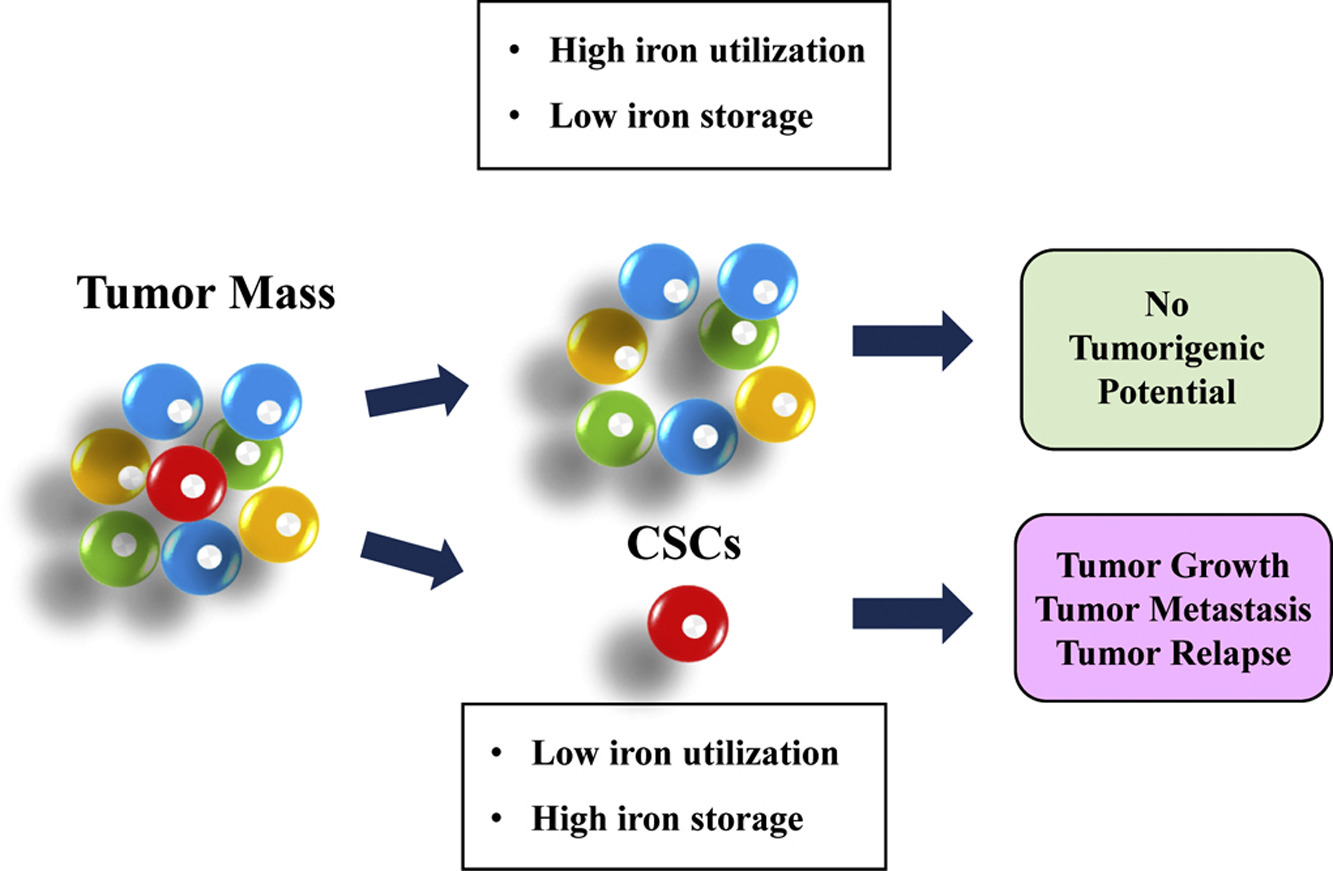
��֢��ϸ����CSC���Ǿ��и�ϸ�������Ե�����ϸ���Ķ�����Ⱥ����Ҫ���ǣ�CSC�����ڵ�ǰ�ı��Ʒ��д���������Ӷ�����ת���Լ������������������������������CSCs������̬�ı仯��ͨ���Ը�ϸ����������Ϊ��������Ҫ���ǣ�����л�쳣�방֢���߸�������������Ͳ���Ԥ����ء�
�방֢��������������һ�£����ǻ��������������Ի�����Ϊ��ҩ�ﻯ;������Ϊ�Ѿ����ǽ������ϼ������������ƣ�����Ŀǰ��Ϊ������ҩ���ܻ�Ӱ��������ٽ�������������������о����·���ǿ����֢�������Ƽ�ֵ��
��

��
��
Dysregulation of iron metabolism in cancer stem cells ...
https://www.sciencedirect.com/science/article/pii/S0891584918312590
The pattern of iron metabolism in CSCs appears to be different from the one
represented in the current model of iron homeostasis in cancer cells, which is
characterized by low iron availability, as the iron provided by increased uptake
and decreased export is used to sustain the high requirement of growing tumor
cells .
Cited by: 5
Publish Year: 2019
Dysregulation of iron metabolism in cancer stem cells
Highlights
•
Cancer stem cells (CSCS) are cells within a tumor with stem cell like features.
•
CSCs are thought to drive tumor growth, metastasis, and chemoresistance.
•
Development of therapies targeted at CSCs holds hope for improved cancer
treatment.
•
Dysregulation of iron homeostasis represents a common feature of cancer stem
cells.
•
Exploiting iron to target CSCs requires better knowledge of iron metabolism in
CSCs.
Abstract
Cancer stem cells (CSCs) are a distinct subpopulation of tumor cells endowed
with stem-like properties. Importantly, CSCs can survive current standard
therapies, resulting in metastatic disease and tumor recurrence. Here we
describe the alterations of iron homeostasis occurring in CSCs, which in general
are characterized by high intracellular iron content. Importantly, abnormalities
of iron metabolism correlate with faster tumor growth and adverse prognosis in
cancer patients.
In line with the dependence of cancer on iron, we also discuss iron-dependent
mechanisms as druggable pathways, as iron chelators have been considered for
tumor therapy and new molecules currently proposed and studied as antineoplastic
drugs may impinge on iron and its capacity to promote oxidative stress to have
therapeutic value in cancer.
��
���ܰ���ϸ����ϸ��������лʧ����
Dysregulation of Iron Metabolism in Cholangiocarcinoma Stem-like Cells
ժҪ
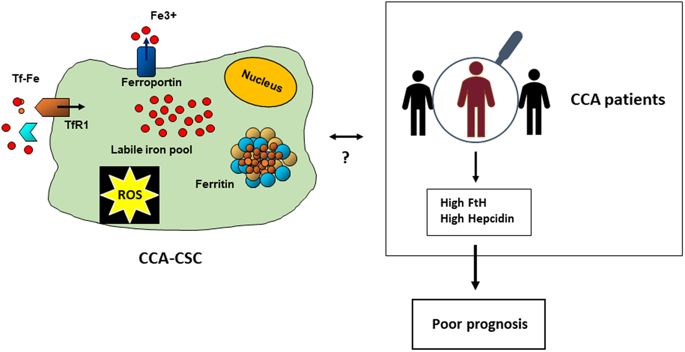
���ܰ���CCA�����ɵ�����Ƥϸ���Ķ���ת������Ļ����Ը���������֢��ϸ����CSC���Ǿ��и�ϸ�������Ե�����ϸ�����Ӽ����������ķ�����������ת���������á����ʵ������£�CSC�γ�3D���壨SPH�����䱣���˾�����������������
��������Ƿ��������IJ�ͬ��������뵥������������ϸ����ȣ�CCA-SPH�����������ӣ�����Ӧ����CSC��־�������ߡ�
��¶�������ϼ�ȥ������DFO)�ɽ���SPH�γ�Ч���Լ�CSC��Ǻ;�������ı������������෴�����á� CCA��Ʒ��n = 104���е���������ʾ�����������Χ�����H�����ף������غ���ת�˵��ױ��オ�ͣ���ת�����������ϵ������⣬���Ƿ���������(ferrin)��������(hepcidin)����������л����Ҫ���ױ���ˮƽ���ߵ�CCA���ߵ�Ԥ�����ڶ�
��Щ���ִ��������ڸ�ϸ����������Ϊһ�ֲ���CCA���������ʹ�л���ӵ����õĵ�һ��֤�ݣ����ܶԸ��õ����Ʒ�����Ӱ�졣
��
1 ���������ŵ�˵��ٴ����о����ģ����������Ըβ����ġ�
2 �����������˹�з�����˹��ѧ���ٴ����顣
3.������ѧ����ҽѧ��ѧϵ���������������
4 ����籾�����籾����������ҽѧ��ѧ��ѧ���\���о��봴�����ġ�
5 �����¿���˹��̹�ն��е���A&M������ѧ����ҽѧ��Scott & White���������о�����
6 �����������Ƥ������ѧ�ڿƺ�ҽѧרҵ��
7 �������������-�ȿƿ���ѧ�ڿƺ���ƹ��������������������ڿƺ����������Ըβ�ѧ����pietro.invernizzi@unimib.it��
8 ������ѧ����ҽѧ��ѧϵ���������������gaetano.cairo@unimi.it��
Dysregulation of Iron Metabolism in Cholangiocarcinoma Stem-like Cells.
Abstract
Cholangiocarcinoma (CCA) is a devastating liver tumour arising from malignant
transformation of bile duct epithelial cells. Cancer stem cells (CSC) are a
subset of tumour cells endowed with stem-like properties, which play a role in
tumour initiation, recurrence and metastasis. In appropriate conditions, CSC
form 3D spheres (SPH), which retain stem-like tumour-initiating features. Here,
we found different expression of iron proteins indicating increased iron
content, oxidative stress and higher expression of CSC markers in CCA-SPH
compared to tumour cells growing as monolayers. Exposure to the iron chelator
desferrioxamine decreased SPH forming efficiency and the expression of CSC
markers and stem-like genes, whereas iron had an opposite effect. Microarray
profiles in CCA samples (n = 104) showed decreased H ferritin, hepcidin and
ferroportin expression in tumours respect to surrounding liver, whereas
transferrin receptor was up-regulated. Moreover, we found a trend toward poorer
outcome in CCA patients with elevated expression of ferritin and hepcidin, two
major proteins of iron metabolism. These findings, which represent the first
evidence of a role for iron in the stem cell compartment as a novel metabolic
factor involved in CCA growth, may have implications for a better therapeutic
approach.
��
һ����ǰ;�İ�֢�����·���������֢��ϸ���е�����л
A promising new approach to cancer therapy: Targeting iron metabolism in cancer stem cells
��
a Institut Necker-Enfants Malades (INEM), Inserm U1151-CNRS UMR 8253, France
b Universit�� Paris Descartes-Sorbonne Paris Cit��, F-75993 Paris, France
Innerm Necker-Enfants Malades��INEM����Inserm U1151-CNRS UMR 8253������b���������ѧ�ѿ�����ѧ-75993�������裬2018��5��18�ս��գ�2018��7��24������2018��7��25�ս��ܣ�2018��7��30�����߿��á�
���Ǵٽ�ϸ����ֳ����������ҪӪ���ء����ǣ��������к�������������ʽ�ͻ�ԭ��ʽ֮��ѭ���������������γ����ɻ������������ɻ��ᵼ��֬�ʹ�����������Ļ�����������Ӧ������DNA������������ӵ����Լ���������ܵĻ���������������������ά������������ת�Ƶ����á����Ļ�ȡ�����ţ�����͵���;�����ڰ�֢���ܵ����ţ����������л���ر��������ϸ��������Ҫ���档������о���ʾ������л�ڰ�֢��ϸ����CSC���е����ã���������CSC������л������������ܸ��ư�֢���ƵĹ�Ч��
��
����ƪ�����У��������ȼ�Ҫ�ܽ�һ�����Ƕ��漰����ϸ���ڹ��̣���ʳ���������ü����방֢֮���ϵ�����⡣����ǿ�������ӡ��������ڰ�֢�е���Ҫ�ԣ��Լ���Щ������ܱ�������ٴ���ʹ�õ������־��Ŀ����ԡ�Ȼ�������ṩ�����ڴ�л�ر�̣���Ƥ-����ת����EMT)�Լ�CSCά�ֺͿ����Աز����ٵı����Ŵ���ǵĵ����е����õĸ��¡�����������������CSCs��������ֵ���������ϸ���������ʴ�֢��������ʽ��DZ����
��
A promising new approach to cancer therapy: Targeting iron metabolism in cancer stem cells
Author links open overlay panelMouradiEl HoutabLeïlaDos
SantosabAhmedHamaïabMaryamMehrpourab a Institut Necker-Enfants Malades (INEM),
Inserm U1151-CNRS UMR 8253, France b Universit�� Paris Descartes-Sorbonne Paris
Cit��, F-75993 Paris, France Received 18 May 2018, Revised 24 July 2018, Accepted
25 July 2018, Available online 30 July 2018. Iron is an essential nutrient that
facilitates cell proliferation and growth. Iron can be detrimental, however. The
ability of iron to cycle between oxidized and reduced forms contributes to the
formation of free radicals. An excess of free radicals leads to lipid
peroxidation, more reactive oxygen species and oxidative stress, damage to DNA
and other biomolecules, and, if potentially, tumorigenesis. Iron also has a role
in the maintenance of the tumor microenvironment and in metastasis. Pathways of
iron acquisition, efflux, storage, and regulation are all perturbed in cancer,
suggesting that reprogramming of iron metabolism is a central aspect of tumor
cell survival. Recent studies have shed light on the role of iron metabolism in
cancer stem cells (CSC) and suggest that specific targeting of iron metabolism
in CSCs may improve the efficacy of cancer therapy. In this review, we first
summarize briefly our current understanding of the intracellular processes
involving iron, the effect of dietary iron, and its relation to cancer. We
emphasize the importance of modifier ��iron genes�� in cancer and the possibility
that these genes may encode biomarkers that may be used clinically. We then
provide an update on the role of iron in metabolic reprogramming, the
epithelial-mesenchymal transition, and the regulation of epigenetic marks
essential for CSC maintenance and plasticity. Finally, we discuss the potential
of targeting a recently discovered form of iron-regulated cell death,
ferroptosis, in CSCs for treatment of cancer.
��
A promising new approach to cancer therapy: Targeting iron metabolism in
cancer stem cells - ScienceDirect
https://www.sciencedirect.com/science/article/pii/S1044579X18300518
��
Frontiers | Iron Metabolism in Liver Cancer Stem Cells ...
https://www.frontiersin.org/articles/10.3389/fonc.2019.00149
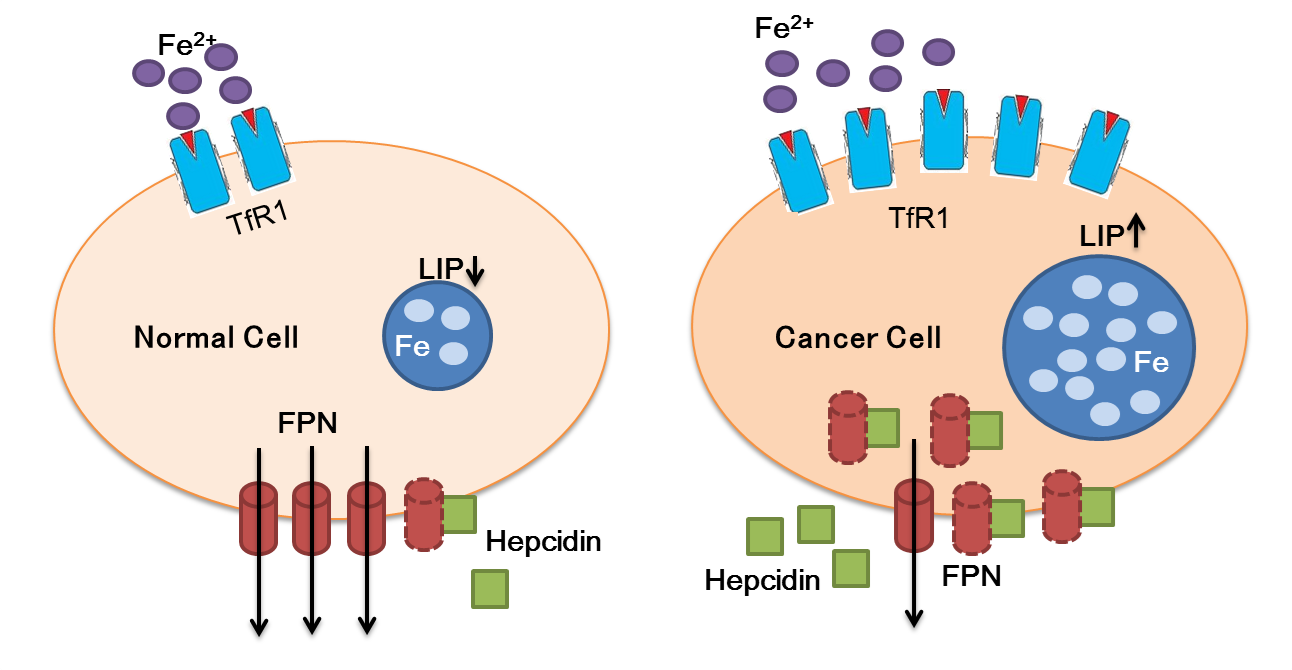
Mar 19, 2019 �� In general, given the high iron needs of tumor cells to sustain
cell proliferation, the alterations of iron trafficking in cancer cells lead to
iron acquisition. To this purpose, cancer cells usually increase iron uptake,
for example by up-regulating TfR1, decrease iron release by inhibiting Fpn, or
both.
Author: Stefania Recalcati, Margherita Correnti, Elena Gammella, Chiara Raggi,
Pietro Invernizzi, Gaetano Ca...
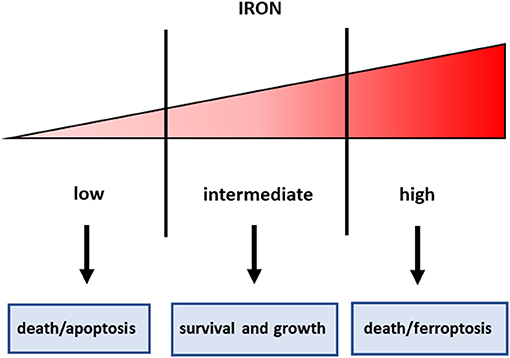
Figure 1. Iron threshold concept. Certain iron levels are required for cell survival and homeostasis, but iron concentrations too low lead to apoptotic cell death, whereas excess iron equally triggers cell death.
��
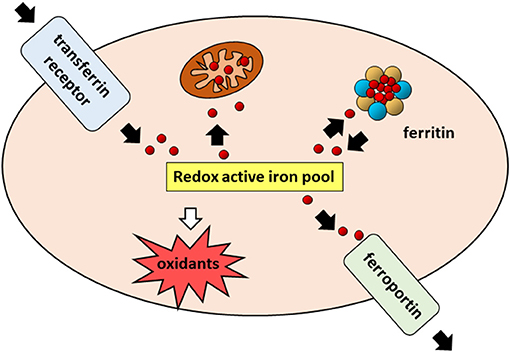
FIGURE 2
www.frontiersin.org
Figure 2. Cellular iron pathways in a nutshell. Transferrin bound iron,
internalized through endocytosis of the transferrin receptor (TfR1), enters a
pool of redox-active iron whose concentration is kept under control by
mechanisms ensuring that the iron which is not used for biochemical processes,
particularly in mitochondria, is either safely stored in cytoplasmic ferritin or
exported by ferroportin.
Publish Year: 2019'
The mechanism of dysregulated iron homeostasis in cancer ...
https://molnut621.wixsite.com/molecularnutrition/single-post/2016/09/08/The-mechanism...
Sep 08, 2016 �� The mechanism of dysregulated iron homeostasis in cancer cells
and the roles of Egr1 and ATF3 in regulation of cancer growth and iron
metabolism September 8, 2016 Medicia Kartawijaya
Author: Medicia Kartawijaya
��
���鶯�������л�����������ڵ��Ŀ��ơ�
Mammalian iron metabolism and its control by iron regulatory proteins��
����
�����ڵ���1��2��IRP1��IRP2��ά��ϸ������̬��
IRP��λ�ڱ���������ȡ�����棬���ú�����ĵ����ʵ�mRNA�Ƿ������е�����ӦԪ����IREs����ϡ��ڹ�ȥ��ʮ���У�������IRP���ͨ���������Ժ��������Ի����Լ�С��IRP2ȱ���IJ����������ȡ�����ش��չ�����漰����ϸ��;��������IREs�ļ���������IRP-IRE��������������̬����Ĺ��̡���IRP���ڵĻ�е������ܻ�Ϊ����л���ҵĻ���������Ҫ�ļ��⡣�����ǡ�����ϸ������ѧ���ؿ���һ���֡�
ǿ��
►IRP1��IRP2�Dz��鶯��ϸ������̬����Ҫ���ڼ��� ►IRP��λ�������գ����棬���ú������ص�mRNA�Ƿ������е�����ӦԪ����IRE����ϡ�
►IRP�������������͵����ʵķ������ء� ►����IREs�ķ��ֽ�ʾ��IRP-IRE��������չ��ϸ������̬֮�ϡ�
►С��IRPȱ�����ƻ�������ƽ�⣬������ѪҺѧ���������Լ����ʹ�л�Լ�����
Mammalian iron metabolism and its control by iron regulatory proteins��
Abstract
Cellular iron homeostasis is maintained by iron regulatory proteins 1 and 2
(IRP1 and IRP2). IRPs bind to iron-responsive elements (IREs) located in the
untranslated regions of mRNAs encoding protein involved in iron uptake, storage,
utilization and export. Over the past decade, significant progress has been made
in understanding how IRPs are regulated by iron-dependent and iron-independent
mechanisms and the pathological consequences of IRP2 deficiency in mice. The
identification of novel IREs involved in diverse cellular pathways has revealed
that the IRP�CIRE network extends to processes other than iron homeostasis. A
mechanistic understanding of IRP regulation will likely yield important insights
into the basis of disorders of iron metabolism. This article is part of a
Special Issue entitled: Cell Biology of Metals.
Highlights
► IRP1 and IRP2 are the principal regulators of mammalian cellular iron
homeostasis. ► IRPs bind to iron-responsive elements (IREs) located in the
untranslated regions of mRNAs involved in iron uptake, storage, utilization and
export. ► IRPs are post-translationally regulated by iron and reactive oxygen
and nitrogen species. ► The identification of novel IREs reveals the presence of
an expanded IRP�CIRE network beyond cellular iron homeostasis. ► IRP deficiency
in mice disrupts iron homeostasis and leads to hematological, neurodegenerative
and metabolic disorders.
Mammalian iron metabolism and its control by iron regulatory proteins -
ScienceDirect
https://www.sciencedirect.com/science/article/pii/S0167488912001267
��
ʳ����״ϸ������ת����������CD71�Ĺ����P����������
Overexpression of transferrin receptor CD71 and its
tumorigenic properties in esophageal squamous cell carcinoma
����˼����
��۴�ѧ
ʳ����״ϸ������ESCC�����������е�������Ҫʳ�ܰ����͡��ڱ��о��У����ǵ�����ת����������CD71��ESCC�е��ٴ���������á�
CD71��ϸ���������о����������ã�������������͵������İ����йء������ǵĶ����У�ʹ�ö����ۺ�ø����Ӧ��61.5���Ļ����м�CD71ת¼����2�����ϵ���������֯��ѧ��������ʾ��ʯ�������������CD71��Ĥ��ϸ���ʾ���ǿ������ֳ��־��Ki-67Ⱦɫƽ��������Ƭ��ʾKi-67Ⱦɫ��ģʽ��CD71�����йء��ٴ��������ݷ���������CD71���������Ϊ����T4�ڵ�ָ�꣨p
=
0.0307������Щ���ݱ���CD71��ESCC֮���к�ǿ����ϵ�����ʹ�ö̸���RNA��siRNA������CD71�������������֤ʵ��ESCC��CD71���°����ԡ���CD71���Ƶ�ϸ���й۲쵽ϸ���������ƺ�S����ϸ��ͣ�͡�DZ�ڵĻ����漰MEK
/ ERK;���ļ���ܶ���֮�����о��ṩ��֤�ݣ���ʾ��CD71��ESCC�е��°��������ٴ�����ԣ����������CD71��ΪESCC�����Ʋ��ԡ�
ת��������һ��Ѫ���ǵ��ף�������ѪҺ��ת����������ϸ�������ת�����������ϣ��Ӷ����������ڻ��������о�������������������ת�����������ڰ������װ����������ٰ����ΰ����ܰ������ڵĶ���ϸ���б��ϵ�[89]
[90] [91]��ת�������ϵ�ҩ�����ϵͳ�ķ�չʹҩ�����ͨ������鵼������������ϸ��[92]�� ...
Kenneth K Y Lai
he University of Hong Kong
Esophageal squamous cell carcinoma (ESCC) is the predominant type of esophageal
cancer in endemic Asian regions. In the present study, we investigated the
clinical implication and role of transferrin receptor CD71 in ESCC. CD71 has a
physiological role in cellular iron intake and is implicated in the
carcinogenesis of various types of tumors. In our cohort, more than a 2-fold
upregulation of the CD71 transcript was detected in 61.5% of patients using
quantitative polymerase chain reaction. Immunohistochemical analysis also showed
strong membranous and cytoplasmic localization of CD71 in paraffin-embedded
tumors. Staining parallel tumor sections with the proliferative marker Ki-67
revealed that the pattern of Ki-67 staining was associated with CD71 expression.
Analysis of clinicopathological data indicated that CD71 overexpression can be
used as an indicator for advanced T4 stage (p=0.0307). These data suggested a
strong link between CD71 and ESCC. Subsequent in vitro assays using short
interfering RNA (siRNA) to suppress CD71 expression confirmed the tumorigenic
properties of CD71 in ESCC; cell growth inhibition and cell cycle arrest at S
phase were observed in CD71-suppressed cells. The underlying mechanism involved
activation of the MEK/ERK pathway. In summary, the present study provides
evidence showing the tumorigenic properties of CD71 in ESCC with clinical
correlations and suggests targeting CD71 as a strategy for the treatment of
ESCC.
Overexpression of transferrin receptor CD71 and its tumorigenic properties in
esophageal squamous cell carcinoma | Request PDF
https://www.researchgate.net/publication/259770754_Overexpression_of_transferrin_receptor_CD71_and_its_tumorigenic_properties_in_esophageal_squamous_cell_carcinoma
��
������֢�Ͱ�ϸ����Ϯ
Iron, inflammation and invasion of cancer cells
������֢������ϸ��������������չΪ��ɢ��֢��ת���йء�ͨ��������������λ����õĸ��ֽ��ʴٽ���Ѫ�����ɴٽ�������ת�ƽ��̡�Ѫ����������������Ҫ�仯����Щ�仯�ɰ�ϸ�����ǻ������¿�ԭ�ʵݸı��������Ѫ�����ɴٽ��IJ����仯������֢��ǿ����֯���ʽ��⣬������Դ��λ�ͷų�����ϸ�������������ϸ���ͷ������ǻ����ı���������¿�ԭ����Tϸ���ʵݵ��¿�ԭ�Ӿ���ת�ƺ���֢�����ɵ�����֢��Ⱦ��ijЩ��Ⱦ�е���֢��������ɢ���棬�������������֢��Ѫ�����ɺ���������֮�����ϵ�����õ��˽�������ϵ���������������ƶ���֢�����Ʒ����²��ԡ�
�ؼ��ʣ�����л����֢�������أ���������
Iron, inflammation and invasion of cancer cells
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4632882
Jul 01, 2015 �� Iron excess is associated with tumorigenesis because it is the
source of mutagenic hydroxyl radical formation that interferes with DNA repair
and affects the signal transduction in cancer cells, acting as a nutrient for
proliferating tumors . The other groups of compounds used for iron-targeted
anti-tumor therapy are the iron chelators.
Cited by: 11
Publish Year: 2015
Author: Eva Fischer-Fodor, Natalia Miklasova, Ioana Berindan-Neagoe, Bhaskar
Saha
Iron, inflammation and invasion of cancer cells
Chronic inflammation is associated with the metastasis of tumor cells evolving
from a benign tumor to disseminating cancer. Such a metastatic progression is
fostered by the angiogenesis propelled by various mediators interacting at the
site of tumor growth. Angiogenesis causes two major changes that are assisted by
altered glycosylation and neo-antigen presentation by the cancer cells. The
angiogenesis-promoted pathological changes include enhanced inflammation and
degradation of tissue matrices releasing tumor cells from the site of its
origin. The degraded tumor cells release the neo-antigens resulting from altered
glycosylation. Presentation of neo-antigens to T cells escalates metastasis and
inflammation. Inflammasome activation and inflammation in several infections are
regulated by iron. Based on the discrete reports, we propose a link between
iron, inflammation, angiogenesis and tumor growth. Knowing the link better may
help us formulate a novel strategy for cancer immunotherapy.
Keywords: iron metabolism, cancer, hepcidin, targeted therapy
��
Depletion of Iron From Tumor Cells Prior To Vitamin C Therapy Quells Cancer
By Bill Sardi
January 2, 2019
donate FacebookTwitterShare
It is widely known that mega-dose vitamin C transiently produces hydrogen
peroxide, an oxidant, to selectively kill cancer cells. In the late 1970s Linus
Pauling and Ewan Cameron were first to report of success utilizing intravenous
vitamin C to produce 1-year survival among 22 percent of otherwise hopeless
cancer patients (chemotherapy at the time was far less effective). Subsequent
studies concluded oral vitamin C could not possibly reach adequate blood
concentrations of vitamin C to produce hydrogen peroxide (cancer cell killing
effect) even though the data from that study ran contrary to the conclusions
drawn. [Knowledge of Health 2016]
Thereafter Steve Hickey and Hilary Roberts posited a dynamic flow theory of
ascorbate (vitamin C) therapy that addressed the fact vitamin C is rapidly
excreted and its oral absorption is drastically reduced when consumed in
mega-doses. Therefore, Hickey and Roberts posed intermittent oral administration
of vitamin C to maintain high blood levels sufficient to exert a cell killing
(cytotoxic) effect in cancer cells. [Journal Orthomolecular Medicine Vol. 20,
2005] Liposomal vitamin C further increases vitamin C delivery inside cells to
potentially effect a cure.
Curing the Incurable: ...
MD JD Thomas E Levy
Best Price: $3.00
Buy New $18.89
(as of 10:30 EDT - Details)
These revelations have led to a renaissance in the use of vitamin C for cancer.
[Knowledge of Health, 2016]
Now another breakthrough in the scientific understanding of vitamin C therapy
for cancer is being reported. Researchers in Japan report extracellular iron
(outside cancer cells) decomposes hydrogen peroxide generated by vitamin C. The
inhibition of cancer cell growth via vitamin C (hydrogen peroxide) is completely
blocked by the presence of iron. [Scientific Reports 2018] This was demonstrated
in leukemia cells (cancer of the blood) in a lab dish. Given that most of the
iron stored in the body is bound to hemoglobin in red blood cells, it is
understandable why iron plays such an important role in leukemia.
The anti-cancer effect of vitamin C is completely abolished by iron. Iron serves
as a growth factor for tumor cells.
With this new understanding, researchers employed iron-binding molecules prior
to vitamin C treatment with demonstrable anti-cancer effects. An iron-chelating
(key-lay-ting) drug, or donation of blood to reduce iron load, or a low iron
diet (in humans, avoidance of red meat) increased the cell-killing effect of
vitamin C.
A reduction of iron combined with vitamin C infusions worked synergistically
(more than additively) to the point where detection and invasion by tumor cells
was completely eliminated. Iron storage (ferritin) was dramatically reduced by
the above measures and then vitamin C-activated hydrogen peroxide killed off
cancer cells readily. Researchers conclude this approach produces a ��novel
anti-leukemic effect.�� [Scientific Reports 2018]
Optimal Nutrition for ...
Thomas E. Levy
Best Price: $2.29
Buy New $13.61
(as of 07:40 EDT - Details)
The idea of utilizing a natural iron chelator, such as IP6 (phytic acid or
phytate) derived from rice bran, prior to oral vitamin C therapy now needs to be
put to the test. The comparative iron chelating properties of various natural
iron chelators is presented in the chart below. It reveals IP6 as a superior
iron chelator even over EDTA which is often employed by clinicians who perform
chelation therapy.
The prospect of home cancer therapy looms as neither IP6 rice bran extract nor
vitamin C are toxic or induce acute side effects. A regimen of IP6 (up to 1600
mg is orally absorbed) followed by a vitamin C chaser is proposed.
The advantage of home vitamin C therapy is that intravenous vitamin C infusions
cannot practically or economically be administered on a daily basis at doctor��s
offices. Cancer rages unchecked in between chemotherapy or even intravenous
vitamin C infusions. Daily oral iron chelation with IP6 rice bran followed by
oral vitamin C therapy (3000 mg vitamin C as ascorbic acid four times a day), or
as an alternative, liposomal vitamin C, may prove to be a safe and novel way of
introducing self-care to cancer therapy. Be aware, home-made mixtures of
lecithin + vitamin C produce emulsified ascorbate but not the microscopic lipid
bi-layered delivery system defined as a liposome. [Whole Foods Magazine Aug.
2018]
Depletion of Iron From Tumor Cells Prior To Vitamin C Therapy - LewRockwell
https://www.lewrockwell.com/2019/01/bill-sardi/depletion-of-iron-from-tumor-cells-prior-to-vitamin-c-therapy-quells-cancer/
��