��
Mast Cell Studies
distributions~gut and air tract, joints,choroid (������Ĥ����liver, artery,
Allergic conditions
Tumor
osteoathritis
cytokines derived~ proinflammatory, allergic response, degradation of tissue,
`histamine, trypatse, heparin, leukotrienes, prostaglandins, chondrontin sulfate, aryl sulfatase, also IL-10
histamine
tryptase��protease from mast cells
��
Mast Cell Biology
Mast cells are tissue resident, granule-containing cells capable of regulating both the innate and adaptive immune response (26). Enrichment of mast cells at environmental interfaces allows these cells to be among the first to respond during pathogen invasion, along with dendritic cells and epithelial cells (27). Moreover, mast cells are typically situated near blood vessels, lymphatics, and nerve endings, enabling them to have long range effects on the host response to pathogens (27, 28). As such, mast cells are critical to immune surveillance, eliciting an immediate reaction to invading pathogens and initiating an appropriate innate and adaptive immune response.
The lung is protected from pathogens by alveolar epithelial cells, endothelial cells, tissue resident alveolar macrophages, dendritic cells, and mast cells.Figure 1. Mast cell activation in response to viral infection. Mast cells are classically known for their response to polyvalent cross-linking of IgE in the FcϵR1 receptor, which is important in protective immunity to helminth worm infection and pathologically associated with allergic disease. However, mast cells also are important tissue sentinel cells for initiating inflammatory response to pathogens. Mast cells can recognize and respond to viruses through several different receptors. These receptors include TLR signaling, such as TLR3 detection of dsRNA, sphingosin-1-phosphate (S1P) binding to its receptor S1PR, and RIG-I recognition of uncapped vRNA. Engagement of these receptors results in mast cell activation leading to immediate degranulation, the de novo synthesis of eicosanoids within minutes of activation, and the de novo synthesis of numerous cytokines, chemokines, and growth factors within hours of activation.
https://www.frontiersin.org/articles/10.3389/fimmu.2015.00238/full
Frontiers | Mast Cell: A Multi-Functional Master Cell | Immunology (frontiersin.org)
��
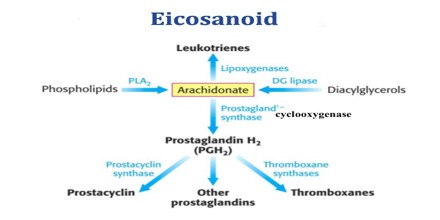
https://www.assignmentpoint.com/science/biology/about-eicosanoid.html
http://tmedweb.tulane.edu/pharmwiki/doku.php/introduction_to_eicosanoids
��
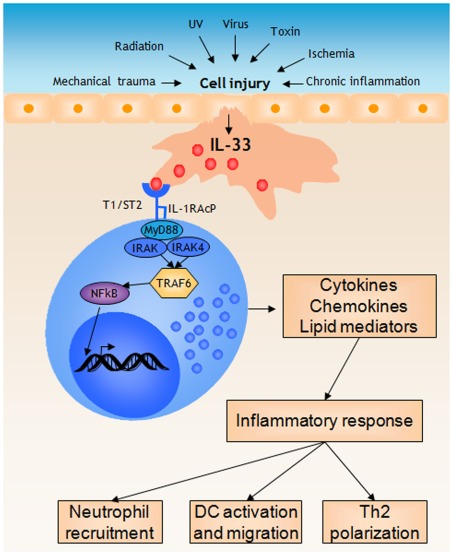
Mast Cells Respond to Cell Injury through the Recognition of IL-33
IL-33 is a member of a big IL-1 cytokine family that regulates innate and adaptive immune systems to promote inflammatory responses [6]. In contrast to the action of IL-1, which is processed and released by live immune cells in response to infection or other triggers, IL-33 acts as an alarmin against injury-induced stress, pathogens, or cell death by activating local immune cells [21,22]. IL-33 is a cytokine with dualfunction. ...
IL-33 is a cytokine that is constitutively expressed in the nuclei of endothelial and epithelial cells [10,11]. The full-length of IL-33, pro-IL-33, serves as a gene regulator that is localized in the nuclei [12], whereas mature IL-33 serves as a cytokine after release into the extracellular space at the onset of tissue injury [13,14]. C-reactive protein (CRP) is an acute-phase protein that serves as an early marker of inflammation or infection. ...
... IL-4, on the other hand, is the signature cytokine of type-2 immunity, IgE-mediated hypersensitivity and can increase proliferation and several MC attributes [22][23][24][25] . IL-33 acts as an "alarmin" released from damaged or injured cells, initiating inflammation, but also Th2-skewed immunity; one of its major target cells is the MC, on which it exerts potent phenotypical and functional effects [26,27] . Finally, retinoic acid (RA) has a crucial function in the skin (both as endogenous hormone and therapeutically), and it is skin MCs that are highly enriched with components of the retinoid network vis-��-vis all major skin cells; consequently skin MCs are potently reshaped by RA [28] . ...
��Mast cells respond to IL-33 released during cell injury by initiating a... | Download Scientific Diagram
https://www.researchgate.net/figure/Mast-cells-respond-to-IL-33-released-during-cell-injury-by-initiating-a-pro-inflammatory_fig2_224919351Mast cells (MCs) are the principal effector cells of IgE-mediated allergy. IL-33 is released by resident skin cells as alarmin upon tissue damage or allergen contact. Owing to their pronounced receptor expression, MCs are important targets of IL-33 action, but consequences for skin MCs are ill-defined, especially upon chronic exposure to IL-33. Mimicking the inflammatory milieu of skin disorders, we found that persistent exposure to IL-33 (over a 5-week period) strengthened skin MC numbers through accelerated cell-cycle progression and restriction of apoptosis. Conversely, IL-33 attenuated degranulation and Fc��RI expression, potentially as a feedback to chronic ��alarmin�� exposure. Interestingly, the negative impact on histamine release was counterbalanced by amplified histamine production. Considering the clinical significance of histamine and scarce information on its regulation, we explored the molecular underpinnings. IL-33 induced swift phosphorylation of p38 and JNK (but not of ERK1/2 or AKT), and stimulated histidine decarboxylase expression. Combining pharmacological inhibition and kinase elimination by Accell-facilitated RNA interference in skin MCs revealed a p38-dependent, but JNK-independent mechanism. Collectively, IL-33 exerts multifaceted effects on cutaneous MCs at a post-maturation stage. The IL-33�Cskin MC axis may contribute to and balance inflammation in chronic skin disorders.
Yin-Yang of IL-33 in Human Skin Mast Cells: Reduced Degranulation, but Augmented Histamine Synthesis through p38 Activation | Request PDF
https://www.researchgate.net/publication/330583564_Yin-Yang_of_IL-33_in_Human_Skin_Mast_Cells_Reduced_Degranulation_but_Augmented_Histamine_Synthesis_through_p38_Activation��
��
IgE-mediated mast cell activation promotes inflammation and cartilage destruction in osteoarthritis
VA Palo Alto Health Care System, United States; Stanford University School of Medicine, United States; University of Padova, Italy; Virginia Commonwealth University School of Medicine, United States
Osteoarthritis is characterized by articular cartilage breakdown, and emerging evidence suggests that dysregulated innate immunity is likely involved. Here, we performed proteomic, transcriptomic, and electron microscopic analyses to demonstrate that mast cells are aberrantly activated in human and murine osteoarthritic joint tissues. Using genetic models of mast cell deficiency, we demonstrate that lack of mast cells attenuates osteoarthritis in mice. Using genetic and pharmacologic approaches, we show that the IgE/Fc��RI/Syk signaling axis is critical for the development of osteoarthritis. We find that mast cell-derived tryptase induces inflammation, chondrocyte apoptosis, and cartilage breakdown. Our findings demonstrate a central role for IgE-dependent mast cell activation in the pathogenesis of osteoarthritis, suggesting that targeting mast cells could provide therapeutic benefit in human osteoarthritis.
Mast cells are sentinels of the innate immune system, poised to rapidly respond to exogenous pathogens and to endogenous danger signals (Bischoff, 2007). A wide variety of stimuli (e.g., allergens that cross-link IgE-bound high affinity IgE receptor (Fc��RI) or antibodies that directly cross-link Fc��RI, cytokines such as IL-33, complement anaphylatoxins, immune complexes, neuropeptides, TLR ligands, etc.) can influence mast cell degranulation and release of pre-formed mediators including histamine, tryptases, pro-inflammatory lipids, cytokines and chemokines (Theoharides et al., 2012; Yu et al., 2016). Importantly, different activation stimuli are capable of inducing distinct mast cell responses in both physiological and pathological settings (Enoksson et al., 2011; Gaudenzio et al., 2016). In allergic disease��a setting in which mast cells have been most extensively studied��these mediators promote chronic allergic inflammation which, if sustained, results in long-term tissue damage, fibrosis, and remodeling (Galli and Tsai, 2012). Similar to the tissue remodeling in allergic diseases, human osteoarthritis and experimental osteoarthritis in rodents are characterized by abnormal and progressive bone and other tissue remodeling (Remst et al., 2014).
IgE-mediated mast cell activation promotes inflammation and cartilage destruction in osteoarthritis | eLife
https://elifesciences.org/articles/39905��
Arch Dermatol Research. 2008 Apr;
Mast cell tryptase and photoaging: possible involvement in the degradation of extra cellular matrix and basement membrane proteins
Arunasiri Iddamalgoda 1, Quang Trong Le, Kenichi Ito, Kiyotaka Tanaka, ollapse
Affiliation
1Department of Research and Development, Ichimaru Pharcos Co. Ltd, Japan.
Mast cells are widely distributed in the connective tissue of the body, but are particularly prominent in tissues such as skin. An increased number of mast cells can be found in the dermis under inflammatory conditions and ultraviolet (UV) exposed skin. Previous investigations have identified matrix metalloproteinases (MMPs) as key enzymes in the degradation of extra cellular matrix (ECM). This study reports about the potential contribution of human mast cell tryptase as a new triggering enzyme in matrix degradation process. Recent studies suggest that mast cell-derived proteases can activate MMPs. We investigated both the degradation of cellular matrix components and activation of MMPs by human tryptase. Mast cells are increased in photoaged skin and the increase of mast cell tryptase in UV irradiated skin was confirmed. Human mast cell tryptase was purified from human tonsils by a series of standard chromatographic procedures. Degradation of collagen type I was achieved by incubation of human type I collagen with tryptase and the fragments were quantified by SDS-PAGE and staining with Coomassie Brilliant Blue 250-R (CBB). Treatment with tryptase resulted in the activation of proMMP-9 as revealed by gelatinolytic activity in type IV collagen zymography. When tryptase was incubated with human type IV collagen, gradual degradation of intact collagen was detected by Western blotting. Furthermore, type IV collagen degradation was observed in the basement membrane (BM) of a three-dimensional (3D) skin model. Degranulation of mast cells, which release tryptase, can activate MMPs and causes direct damage to ECM proteins. These findings strongly implicate that tryptase either alone or in conjunction with activation of MMPs, can participate in ECM damage and the possible destruction of BM leading to photoaging.��
FASEB J. 2020 Aug
A role for mast cells in geographic atrophy
Mast cells (MCs) are the initial responders of innate immunity and their degranulation contribute to various etiologies. While the abundance of MCs in the choroid implies their fundamental importance in the eye, little is known about the significance of MCs and their degranulation in choroid. The cause of geographic atrophy (GA), a progressive dry form of age-related macular degeneration is elusive and there is currently no therapy for this blinding disorder. Here we demonstrate in both human GA and a rat model for GA, that MC degranulation and MC-derived tryptase are central to disease progression. Retinal pigment epithelium degeneration followed by retinal and choroidal thinning, characteristic phenotypes of GA, were driven by continuous choroidal MC stimulation and activation in a slow release fashion in the rat. Genetic manipulation of MCs, pharmacological intervention targeting MC degranulation with ketotifen fumarate or inhibition of MC-derived tryptase with APC 366 prevented all of GA-like phenotypes following MC degranulation in the rat model. Our results demonstrate the fundamental role of choroidal MC involvement in GA disease etiology, and will provide new opportunities for understanding GA pathology and identifying novel therapies targeting MCs.A role for mast cells in geographic atrophy - PubMed (nih.gov)
��
Arterioscler Thromb Vasc Biology. 1998 Nov;
Activation of matrix-degrading metalloproteinases by mast cell proteases in atherosclerotic plaques
Bristol Heart Institute, University of Bristol, Bristol Royal Infirmary, Bristol, UKRecent studies suggest that mast cell-derived neutral proteases can activate matrix-degrading metalloproteinases (MMPs). We have investigated the role of the mast cell proteases tryptase and chymase in the activation of MMPs in human carotid endarterectomy specimens (atherosclerotic, n=32) and postmortem carotid arteries (control, n=17). In vitro degranulation of mast cells in atherosclerotic carotid arteries by compound 48/80 caused a significant increase in MMP activity. Addition of the nonselective tryptase inhibitor antipain, the specific trypsinlike protease inhibitor 4-amidinophenylmethanesulfonyl fluoride, and the chymase inhibitor chymostatin reduced this increase in MMP activity by 30+/-6%, 23+/-6%, and 9+/-2%, respectively. Immunocytochemistry identified significantly higher numbers of tryptase-containing cells (mast cells) and cells expressing MMP-1 and MMP-3 in the "shoulder" regions of atherosclerotic artery lesions compared with the tunica media of control arteries. Dual immunocytochemistry showed collocation of MMP-1 and MMP-3 with mast cells in the shoulder regions. Degranulation was observed in 78+/-5% (mean+/-SEM) of mast cells in this area, whereas nonactivated mast cells were observed in all other areas. In situ zymography revealed caseinolytic and gelatinolytic activity in these areas. In conclusion, in vitro mast cell degranulation, which releases mast cell proteases, in carotid arteries increases MMP activity. Furthermore, elevated MMP-1 and MMP-3 expression is collocated with increased numbers of degranulated mast cells and with greater MMP activity in the shoulder regions of atherosclerotic plaques. Activation of MMPs by mast cell-derived proteases may be an important mechanism in atherosclerotic plaque destabilization.
��
Effects of essential fatty acids on mediators of mast cells in culture
The objective of this study was to investigate the effects of ��-linolenic acid (18:3n-3) and linoleic acid (18:2n-6) on the fatty acid composition and the activity and release of mast cell mediators in the canine mastocytoma cell line C2. Cells were cultured in Dulbecco's modified Eagle's medium mixed with 50% Ham's F12 (containing linoleic acid 0.14 ��M). The basic medium (DEH) was supplemented with 0.14 ��M ��-linolenic acid. 14.0 ��M ��-linolenic acid (DEH-n-3) or 14.0 ��M linoleic acid (DEH-n-6) was added. Eight days after culturing of C2 in DEH-n-3 we measured elevated levels of n-3 fatty acids up to 22:3. The tryptase activity and the stimulated PGE2 production and histamine release were reduced. In contrast, after culturing of C2 in DEH-n-6 we determined elevated levels of n-6 fatty acids up to 20:3, increased tryptase activity and stimulated histamine release. Thus 18:3n-3 has anti-inflammatory effects in cultured canine mastocytoma cells.����֬���������Էʴ�ϸ�����ʵ�Ӱ��
�����о���Ŀ�����о���-�����ᣨ18��3n-3���������ᣨ18��2n-6����Ȯ�ʴ�ϸ����ϸ��ϵC2��֬��������Լ��ʴ�ϸ�����ʵĻ��Ժ��ͷŵ�Ӱ�졣��ϸ������50��Ham's F12����0.14��M�����ᣩ��ϵ�Dulbecco's����Eagle��������������������������DEH��������0.14��M ��-�����ᡣ����14.0�� M�Ħ�-�����ᣨDEH-n-3����14.0��M�������ᣨDEH-n-6������DEH-n-3������C2��������Dz������ߴ�22��3��n-3֬����ˮƽ���ߡ����������ȵ���ø�Ļ����Լ��̼���PGE2�IJ������鰷���ͷš��෴����DEH-n-6������C2������ȷ���˸ߴ�20��3��n-6֬����ˮƽ���ߣ����ȵ���ø�������Ӻ��鰷�ͷ��ܵ��̼�����ˣ�18��3n-3��������Ȯ�ʴ�ϸ����ϸ���о��п������á�
Effects of essential fatty acids on mediators of mast cells in culture - ScienceDirect
https://www.sciencedirect.com/science/article/abs/pii/S095232780300022X��
Mast Cell Interleukin-10 Drives Localized Tolerance in ...
https://www.cell.com/immunity/fulltext/S1074-7613(13)00050-2
Mast Cell Interleukin-10 Drives Localized Tolerance in Chronic Bladder Infection. Previous Article Human Inflammatory Dendritic Cells Induce Th17 Cell Differentiation. Next Article Interleukin-9 Is Required for Allergic Airway Inflammation Mediated by the Cytokine TSLP. Summary.
Cited by: 140
Publish Year: 2013
Author: Cheryl Y. Chan, Ashley L. St. John, Soman N. Abraham
Induction of Interleukin-10 Producing Dendritic Cells As a ...
https://www.ncbi.nlm.nih.gov/pubmed/29616018
Mar 19, 2018 �� Among the cytokines produced by tolerogenic DCs, interleukin 10 (IL-10) is a key regulatory cytokine limiting und ultimately terminating excessive T-cell responses to microbial pathogens to prevent chronic inflammation and tissue damage.